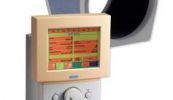
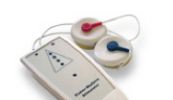
QLASER

- ‹ vorige
- 805 van de 1068
- volgende ›
http://www.craigzunkadds.com/our-services/low-level-laser ![]()
Low Level Laser therapy is a treatment in which Subtle Energy . . . . .
. Low level lasers like the Q Laser Series use less than one Watt of power and they produce what can be best described as a "Healing Light". Low level lasers have been noted as being a catalyst for
Benificial Healing.
04/2018 https://www.devicewatch.org/reg/lytle.shtml ![]()
Quack Device (Qlaser) Marketers Get Prison Sentences
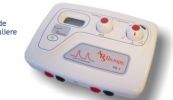
Een van de vele LASER-apparaten deze QLASER.
Geen enkele laser heeft "subtle energy"; ze stralen licht uit. Heel weinig zoals bij deze, of heel veel als bij lasersnijmachines ![]() .
.
Iedereen maakt zich wel eens zorg. Dus gelijk maar een Qlaser aanschaffen ?
Niet doen, want of het ervaren van de Qlaser daadwerkelijk iets aan de kwalen doet staat er niet.
FDA approved the Qlaser1000 for Osteoarthritis of the Hand.
01/ 2018 12 jaar cell voor consumentenbedrog
(Bron: Consumer Health Digest #18-17, April 29, 2018 )
In January, Robert "Larry" Lytle, who organized and led a scheme to deceptively promote and sell the QLaser system as effective for treating more than two dozen health problems, pleaded guilty to one count of conspiracy to introduce misbranded medical devices into interstate commerce with the intent to defraud and mislead, and one count of criminal contempt. As part of his plea agreement:
Lytle admitted that beginning in 2005 he entered into an agreement with others to sell medical devices with false and misleading labeling in order to defraud consumers and that he continued to do so in violation of a federal court order.
Lytle acknowledged that he obtained at least $16,669,015 over the course of the scheme.
Lytle made an initial restitution payment of $637,000 and turned over several thousand dollars' worth of gold and silver coins to be applied to restitution.
He received one of the longest prison sentences ever imposed on a device marketer who has made outrageous health claims: 12 years in prison, followed by 2 years of supervised release.
================
Reclame even checken:
http://www.accessdata.fda.gov/cdrh_docs/pdf8/K080513.pdf ![]() leert dat het slechts om een "clearance" gaat. En er is een extra conditie aan verbonden:
leert dat het slechts om een "clearance" gaat. En er is een extra conditie aan verbonden:
“The QLaser system is indicated for providing temporary relief of pain associated with osteoarthritis of the hand, which has been diagnosed by a physician or other licensed medical professional. No other medical treatment claims are made or implied.”
Verder doet Qlaser te scherp zaken:
http://www.fda.gov/ICECI/EnforcementActions/WarningLetters/ucm246619.html ![]()