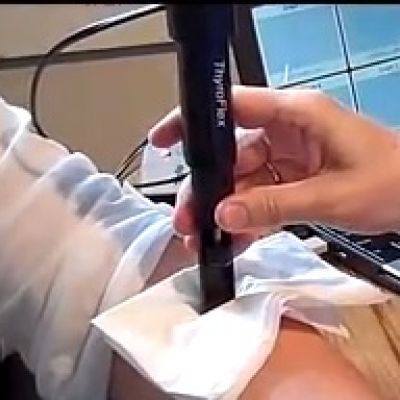
THYROFLEX
http://drcherylkasdorf.com/2012/01/16/tired-of-being-tired/ ![]()
Are you one of the 30% of the U.S. population that has thyroid dysfunction?
. . .
Due to government and FDA regulations the following must be stated: These statements have not been evaluated by the Food and Drug Administration.
The information provided is not a substitute for professional medical opinion.
De bovenstaande waarschuwing blijkt 07/2012 verdwenen. De mededeling over FDA-approval klopt niet want die luidt:
https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfrl/ldetails.cfm?lid=413599 ![]()
Iets wat anderen ook opmerkten
https://sciencebasedmedicine.org/how-to-regulate-cam-aussie-style/ ![]()
Websites claim the Thyroflex is 98.5% accurate. The Thyroflex is actually approved by the FDA, as pointed out by one website. However, it is listed as a neurological diagnostic device. Perhaps the FDA should have been tipped off by the name that it wasn’t going to be used that way.
WAARSCHUWING:
Consumer Health Digest #18-39
September 30, 2018
The device is not cleared or approved by the U.S. Food and Drug Administration. It is merely registered as a percussion instrument, which the FDA classifies as a neurological diagnostic device that is exempt from premarket notification procedures as it is "used . . . to provide light blows to a body part," as "a diagnostic aid during physical examinations." However, NiTek's claims go beyond what is permissible for exempt devices. [Barrett, S. A skeptical look at Thyroflex testing. Quackwatch. Sept 25, 2018]
http://www.quackwatch.org/01QuackeryRelatedTopics/Tests/thyroflex/overview.html ![]()
For all of the above reasons, I believe it would be wise to avoid Thyroflex testing and those who recommend it.
http://www.thyroidinstitute.org/innovative_technology_thyroflex.html ![]()
The clinical trials were evaluated by our statistician and will have been submitted for publishing in a leading medical journal.The Device will move to the final stage of approval as a class II medical device by the FDA, in conjunction with a major medical facility.
http://www.prweb.com/releases/2015/02/prweb12547313.htm ![]()
. . . . . in Kettering and Mason, Ohio has acquired central and southwest Ohio’s first Thyroflex machine, which tests women’s thyroid levels to determine dosage for prescription thyroid medicine.
The Thyroflex tests women’s thyroid levels with 98.5 to 99 percent accuracy. It is FDA approved.
Die FDA-approval is dus duidelijk niet in orde.
De test zelf waarschijnlijk ook niet. Zie het eerdere commentaar in:
https://sciencebasedmedicine.org/how-to-regulate-cam-aussie-style/ ![]()